/GettyImages-548001483-56a133965f9b58b7d0bcfd01.jpg)
There are various versions of this basic setup that may be employed. For astronomical purposes, the telescope must be equipped with the light dispersion device. Most spectroscopic analysis in the laboratory starts with a sample to be analyzed, then a light source is chosen from any desired range of the light spectrum, then the light goes through the sample to a dispersion array (diffraction grating instrument) and is captured by a photodiode. Spectral measurement devices are referred to as spectrometers, spectrophotometers, spectrographs or spectral analyzers. Spectroscopy is a branch of science concerned with the spectra of electromagnetic radiation as a function of its wavelength or frequency measured by spectrographic equipment, and other techniques, in order to obtain information concerning the structure and properties of matter. 
Important applications include biomedical spectroscopy in the areas of tissue analysis and medical imaging. Spectroscopy, primarily in the electromagnetic spectrum, is a fundamental exploratory tool in the fields of astronomy, chemistry, materials science, and physics, allowing the composition, physical structure and electronic structure of matter to be investigated at the atomic, molecular and macro scale, and over astronomical distances. Historically, spectroscopy originated as the study of the wavelength dependence of the absorption by gas phase matter of visible light dispersed by a prism. In simpler terms, spectroscopy is the precise study of color as generalized from visible light to all bands of the electromagnetic spectrum. Matter waves and acoustic waves can also be considered forms of radiative energy, and recently gravitational waves have been associated with a spectral signature in the context of the Laser Interferometer Gravitational-Wave Observatory (LIGO). Spectroscopy is the field of study that measures and interprets the electromagnetic spectra that result from the interaction between electromagnetic radiation and matter as a function of the wavelength or frequency of the radiation. The expression forbidden transitions is often used, but this does not mean that these transitions cannot occur, only that they are electric-dipole-forbidden.An example of spectroscopy: a prism analyses white light by dispersing it into its component colors. These considerations generate different sets of transitions rules depending on the multipole order and type. Thus, parity does not change for E-even or M-odd multipoles, while it changes for E-odd or M-even multipoles. In quantum mechanics the basis for a spectroscopic selection rule is the value of the transition moment integral ∫ ψ 1 ∗ μ ψ 2 d τ, In the following, mainly atomic and molecular transitions are considered. 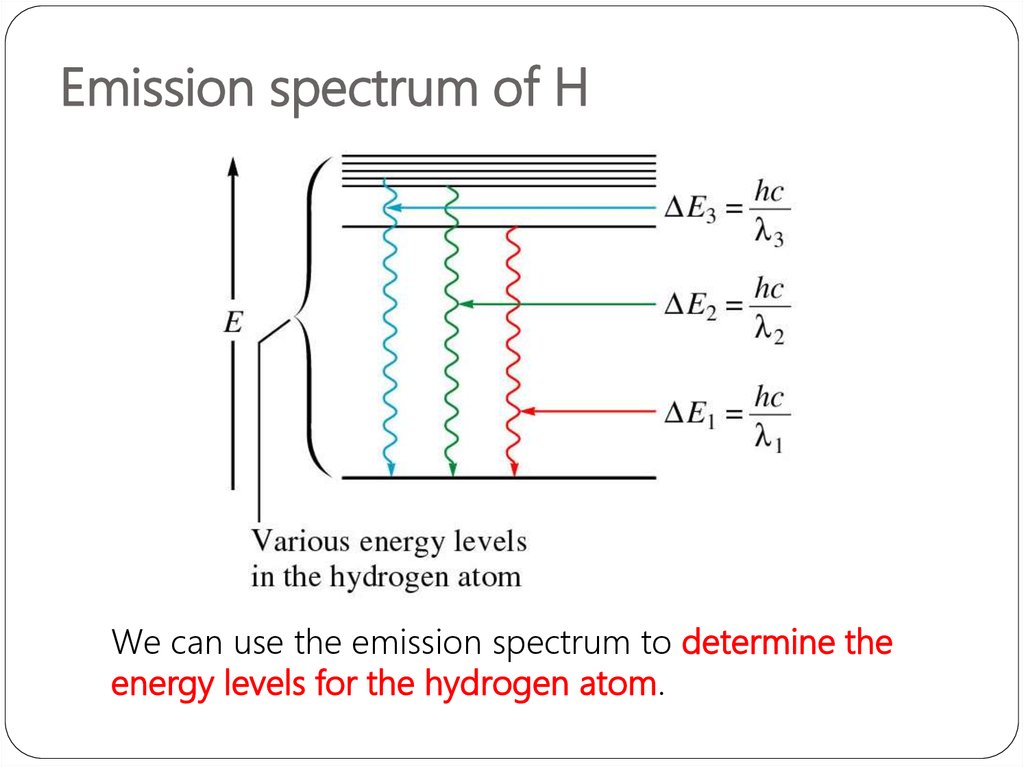 
The selection rule also plays a role in chemical reactions, where some are formally spin-forbidden reactions, that is, reactions where the spin state changes at least once from reactants to products. The selection rules may differ according to the technique used to observe the transition. Selection rules have been derived for electromagnetic transitions in molecules, in atoms, in atomic nuclei, and so on. In physics and chemistry, a selection rule, or transition rule, formally constrains the possible transitions of a system from one quantum state to another. Formal constraint on the possible transitions of a system from one quantum state to another
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
